 |
| Mareva Fevre, IBM Research-Almaden |
 |
|
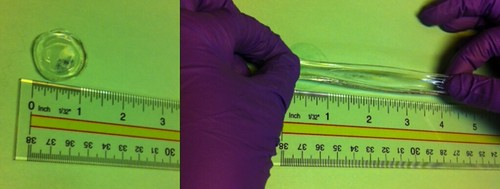
The
elasticity of our organogel on display
|
 |
|
IBM’s gel
(B), once heated, returns to its original state demonstrating
recyclable, remoldable properties. A typical gel (A) retains its form with heat. |
Labels: ibm research almaden, nanotechnology, organogel, polymer